Carlos Mavioso, Ricardo J. Araújo, H elder P. Oliveira, João C. Anacleto, Maria Antónia Vasconcelos, David Pinto, Pedro F. Gouveia, Celeste Alves, Fátima Cardoso, Jaime S. Cardoso, Maria João Cardoso
KEYWORDS
Microsurgery. Perforators. Pre-operative mapping. DIEP. Flap. Automatic detection. Computer vision. Image analysis.
ABSTRACT
The deep inferior epigastric perforator (DIEP) is the most commonly used free flap in mastectomy reconstruction.
Preoperative imaging techniques are routinely used to detect location, diameter and course of perforators, with direct intervention from the imaging team, who subsequently draw a chart that will help surgeons choosing the best vascular support for the reconstruction.
In this work, the feasibility of using a computer software to support the preoperative planning of 40 patients proposed for breast reconstruction with a DIEP flap is evaluated for the first time. Blood vessel centreline extraction and local characterization algorithms are applied to identify perforators and compared with the manual mapping, aiming to reduce the time spent by the imaging team, as well as the inherent subjectivity to the task.
Comparing with the measures taken during surgery, the software calibre estimates were worse for vessels smaller than 1.5 mm (P . 6e-4) but better for the remaining ones (P . 2e-3). Regarding vessel location, the vertical component of the software output was significantly different from the manual measure (P . 0.02), nonetheless that was irrelevant during surgery as errors in the order of 2e3 mm do not have impact in the dissection step.
Our trials support that a reduction of the time spent is achievable using the automatic tool (about 2 h/case).
The introduction of artificial intelligence in clinical practice intends to simplify the work of health professionals and to provide better outcomes to patients. This pilot study paves the way for a success story.
1. INTRODUCTION
The deep inferior epigastric perforator (DIEP) is the most commonly used free flap in autologous reconstruction for mastectomy, either immediate or delayed.
Allen and Treece [1] independently performed the first successful DIEP flap for breast reconstruction in 1992. The typical indication was a fit patient, proposed for a delayed reconstruction after a previous unilateral mastectomy, with enough tissue on the lower abdomen to reconstruct the absent breast. By then, perforators identification was done intra-operatively by the surgical team. Indications for free flap reconstruction underwent a considerable evolution over the last twenty years, and actually DIEP flaps are frequently done in immediate reconstruction, uni- or bilaterally, together with classic mastectomy and skin sparing techniques, and different skin approaches (like reduction patterns), either for cancer or for prophylactic reasons. Patient selection criteria also became less strict (e.g. diabetic, smokers), and several technical improvements have been progressively introduced [2].
One of the most important technical achievements was the pre-operative identification of the infra-umbilical perforator vessels. The high variability of the perforator’s location, and the time spent by the surgical team identifying “the best perforator(s)” brought imaging techniques to the arena to try to help the mapping of the existent perforators before surgery, allowing not only a more adequate planning, but also a reduction in the duration of surgery [3].
Three imaging methods are currently used to map the perforator vessels before surgery: the Doppler Ultrasound (DUS) [4]; the Computed Tomography Angiography (Angio CT) and the Magnetic Resonance Imaging Angiography (Angio MRI) [3].
Angio CT and Angio MRI are more reproducible than DUS, and in both cases the perforators are identified by the imaging team and a “map” is drawn to help the surgeon in identifying the best vascular anatomy for the reconstruction. This process is time consuming and includes two phases: technician dependent – the slice reconstruction and the vessel identification; radiologist dependent – the manual identification of each of the contrast enhanced perforating branches of the epigastric, their location in the abdomen related to the umbilicus, calibre and direction after perforating the abdominal fascia (Fig. 1).
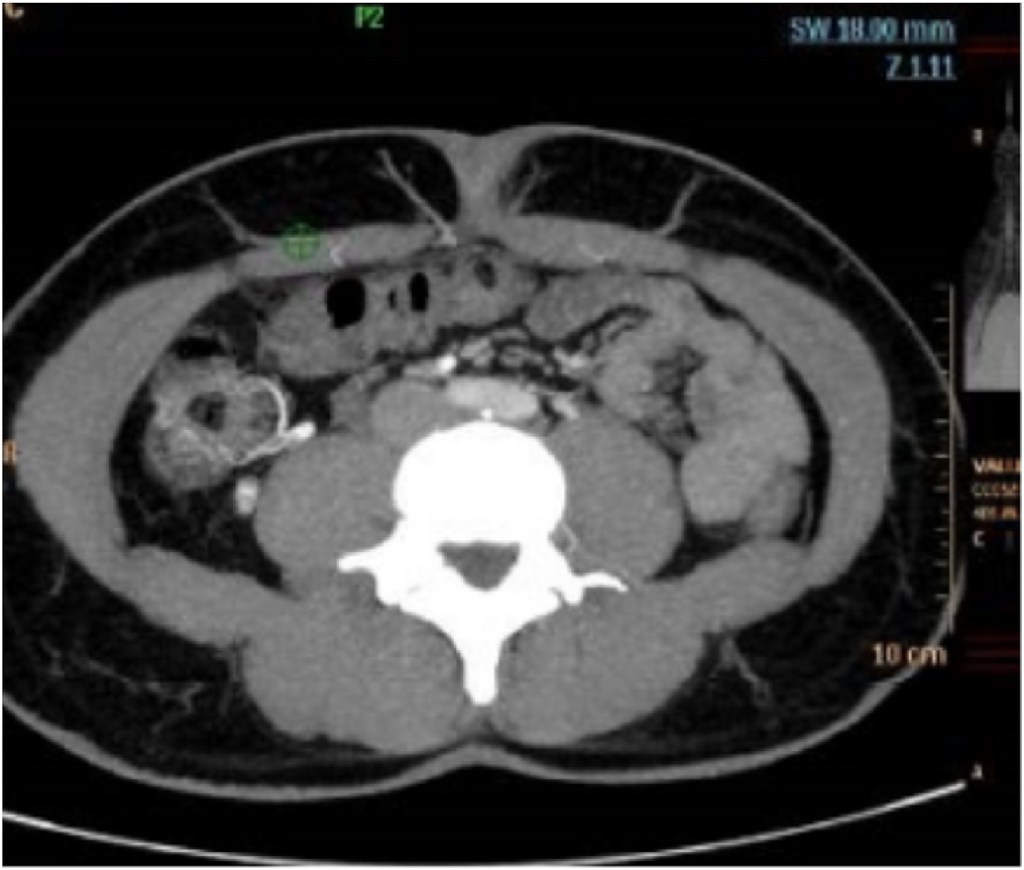
view).
Even with the recent technological developments in Angio CT data acquisition, the spacing between voxels usually does not go below 0.7 mm. Identified perforators commonly have calibres between 1 and 3 mm; often the signal of a perforator cross section spreads across 2 or 3 voxels. Additionally, the perforator’s course pierces different tissues: the rectus abdominis muscle and the subcutaneous fat. In Angio CT, muscle regions also show high radiodensity, making perforators signal usually harder to distinguish in their intramuscular region.
Software tools to enhance identification and characterization of perforators have been proposed in the past. VirSSPA, for example, is a virtual reality tool allowing the imaging team to show results of their self-analysis to the surgical team in a 3D environment [5]. In another study, a plugin was developed to make the delineation of perforators and their characterization easier [6]. However, none of these tools was able to segment the vessels automatically.
Important works have been developed in the retina [7] and coronary arteries [8], but a thorough search of relevant literature revealed that no work has been done so far in the automatic identification of perforators for microsurgery.
We are presenting this pilot study that aims to reduce duration and subjectivity of the pre-operative Angio CT imaging teamwork, using a semi-automatic methodology [9] relying on Computer Vision techniques to map perforator branches for surgical guidance. This way we intend to achieve optimal patient outcome with minimal time consumption.
2. MATERIAL AND METHODS
Forty patients proposed for uni (35) or bilateral [5] breast reconstruction, either immediate or delayed, were included after signing the informed consent to participate in the current study.
An Angio CT was performed pre-operatively in the 40 included patients, using a CT Spectral Scanner with a 64-detector row (Philips Iqon), a 140 Kv tube voltage and a 120e160 mAs fixed tube current. Further settings included 0.75 rotation time, collimation of 128, pitch of 0.609 and a 512 Matrix. 80e100 ml of contrast was injected with images acquired in Bolus Tracking. Images started 5 cm above the umbilicus to the lesser trochanter (range of 32 cm) with a thickness of 0.8 mm and an increment of 0.4 mm.
2.1. Manual identification
For each of the 40 patients, the exams were reconstructed and annotated by the same imaging team and a map report was produced with the calibre, orientation and distance to the umbilicus (horizontally and vertically). Accordingly to the information given by the imaging technicians, around 60e90 min are spent with each case. The validation of the reconstructed images and report creation by the radiologist also takes around 60e90 min, making the complete evaluation of the radiology team last 2e3 h for each case, depending on the number or perforators and particular characteristics of each patient.
2.2. Semi-automatic identification
Anonymized images were blindly analysed by the engineering team using a divide-and-conquer methodology [9], combining a tracking procedure and a minimum cost approach (Figs. 2 and 3). A map report was produced at the end of the process, reporting the same features of the manual identification: calibre, orientation and distance to the umbilicus (horizontally and vertically).
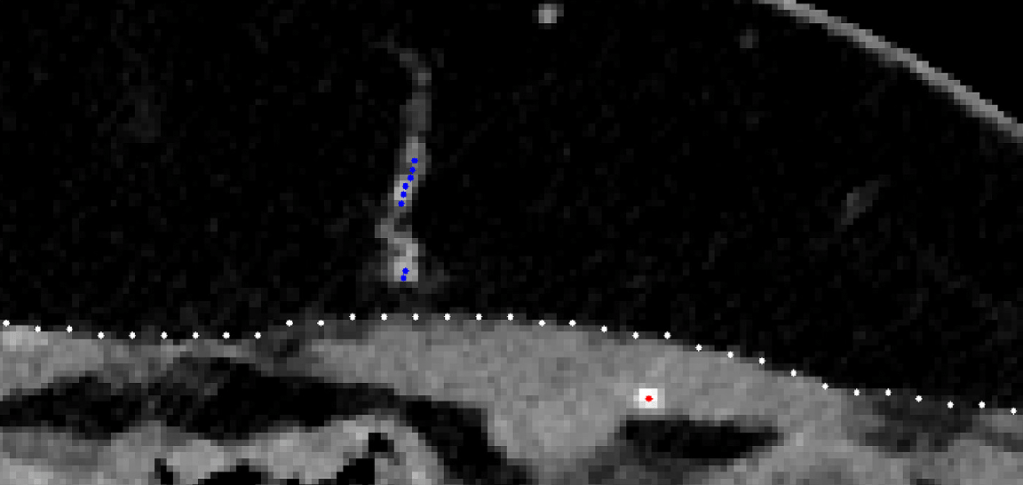
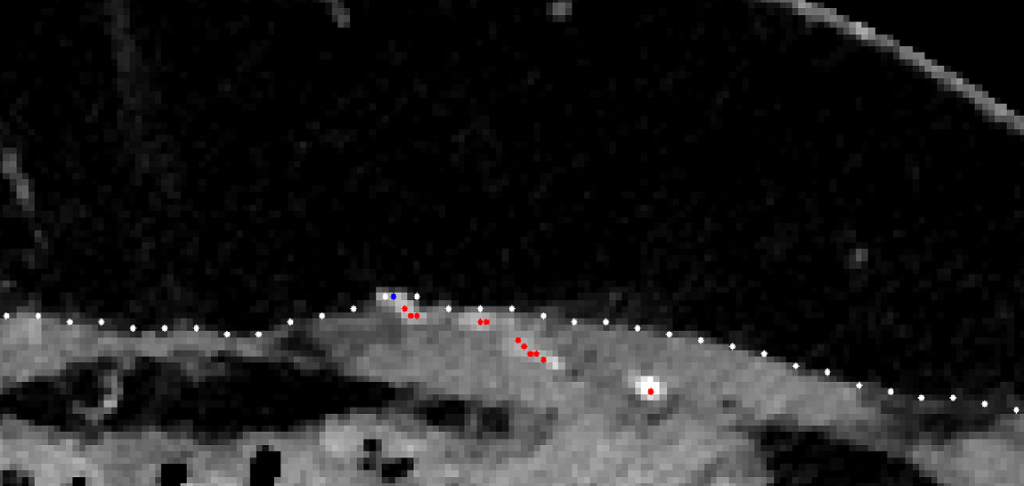
The tracking procedure extracts the subcutaneous course of a perforator, from a point near its end to the location where it leaves the rectus abdominis fascia. The minimum cost approach finds the intramuscular path from this last point to the location where the artery perforates the posterior lamella of the rectus abdominis muscle. The process lasts around 30 min (mostly due to the time spent loading the data and selecting the few points required).
2.3. Intra-operative marking
The two independent maps produced for each case (manually and automatically) were marked intra-operatively on the abdomen of the patients using the horizontal and vertical distances to the umbilicus with different colours (Fig. 4). During surgery, each vessel was checked, and its characteristics annotated. This intraoperative information was used as the ground truth.
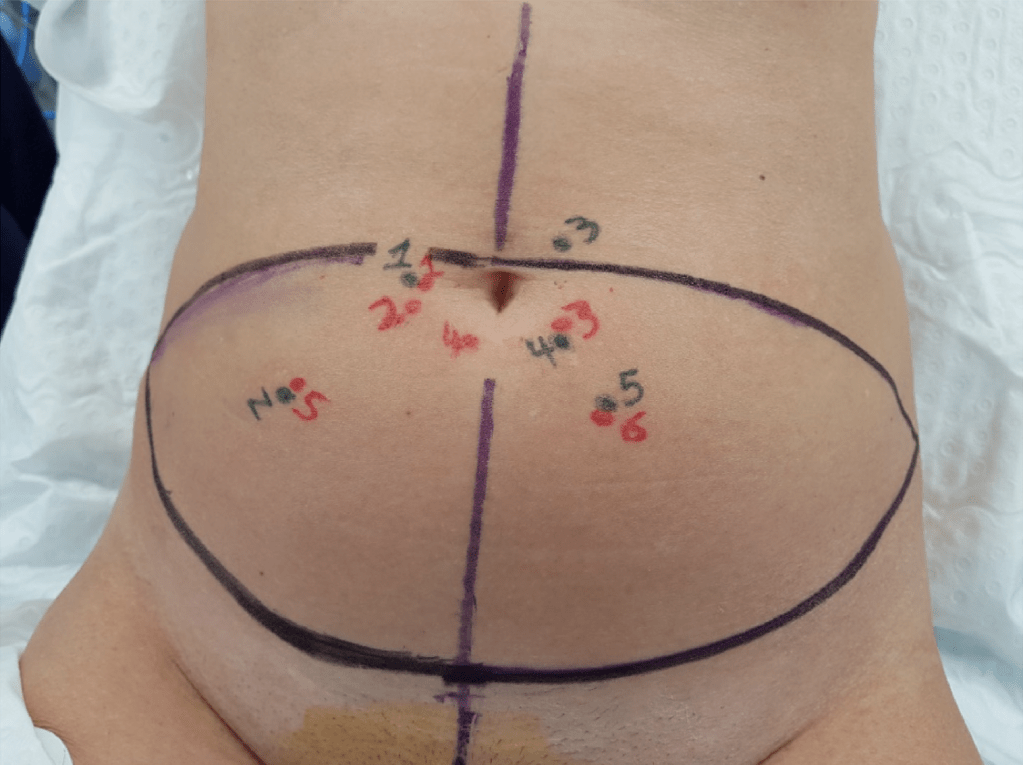
references to colour in this figure legend, the reader is referred to the Web version of this article.)
2.4. Statistical analysis
1) A paired sample t-test with a significance level alpha = 0:05 evaluated the difference in calibres between both methods (null hypothesis H0 was that the average of the differences is 0, and the alternative hypothesis H1 is that this does not hold). The same analysis was also performed only in the perforators that ended up being included in the flaps by the surgical team. Effect size was measured through the standardized mean difference and interpreted accordingly (estimates of 0.20, 0.50, and 0.80 denote, respectively, a small, medium, and large effect size) (14).
2) A Wilcoxon rank-signed test with a significance level alpha = 0:05 was used to access the difference in calibre estimation between both methods when compared to calibres reported by the surgical team intra-operatively (ground truth). This analysis was performed in perforators having a calibre larger than 1.5 mm (selected frequently) and also in the ones having 1.5 mm or less (selected infrequently). Effect size was measured through correlation and interpreted accordingly (estimates of 0.10, 0.30, and 0.50 denote, respectively, a small, medium, and large effect size) (14).
3) Finally, the software was evaluated on how well it estimated the localization of perforators where they leave the fascia of the rectus abdominis muscle. The errors of the software were divided into vertical and horizontal errors and a one sample test was performed for each of them, where in H0 the average error is 0 and in H1 it is not 0. Effect size was measured through the standardized mean difference. Here, the true locations of the perforators were considered to be the ones reported by the radiological team, as these measures cannot be accurately obtained during the surgery. Additionally, the subjectivity is practically negligible for this parameter, in contrast with calibre measurement.
3. RESULTS
From the 40 Angio CTs, 180 perforators have been manually identified pre-operatively by the imaging team, and 183 by the image processing algorithm. A total of 234 vessels were confirmed intra-operatively (ground-truth). From those, a total of 129 vessels were identified simultaneously by both methods.
A statistically significant difference was detected when estimating calibres using the manual and software methods, P ≈ 1e-5 (128 of freedom), and the effect size was medium, r ≈ 0.39. However, the paired sample t-test revealed no statistically significant difference when estimating the calibres of the perforators selected for the flap using the manual and software methods, P ≈ 0.44 (40 of freedom), and the effect size was also small, r ≈ 0.12. Fig. 5a and b shows the distribution of the differences between the calibre estimates of both methods when considering, respectively, all the perforators or only the ones included in the flaps.
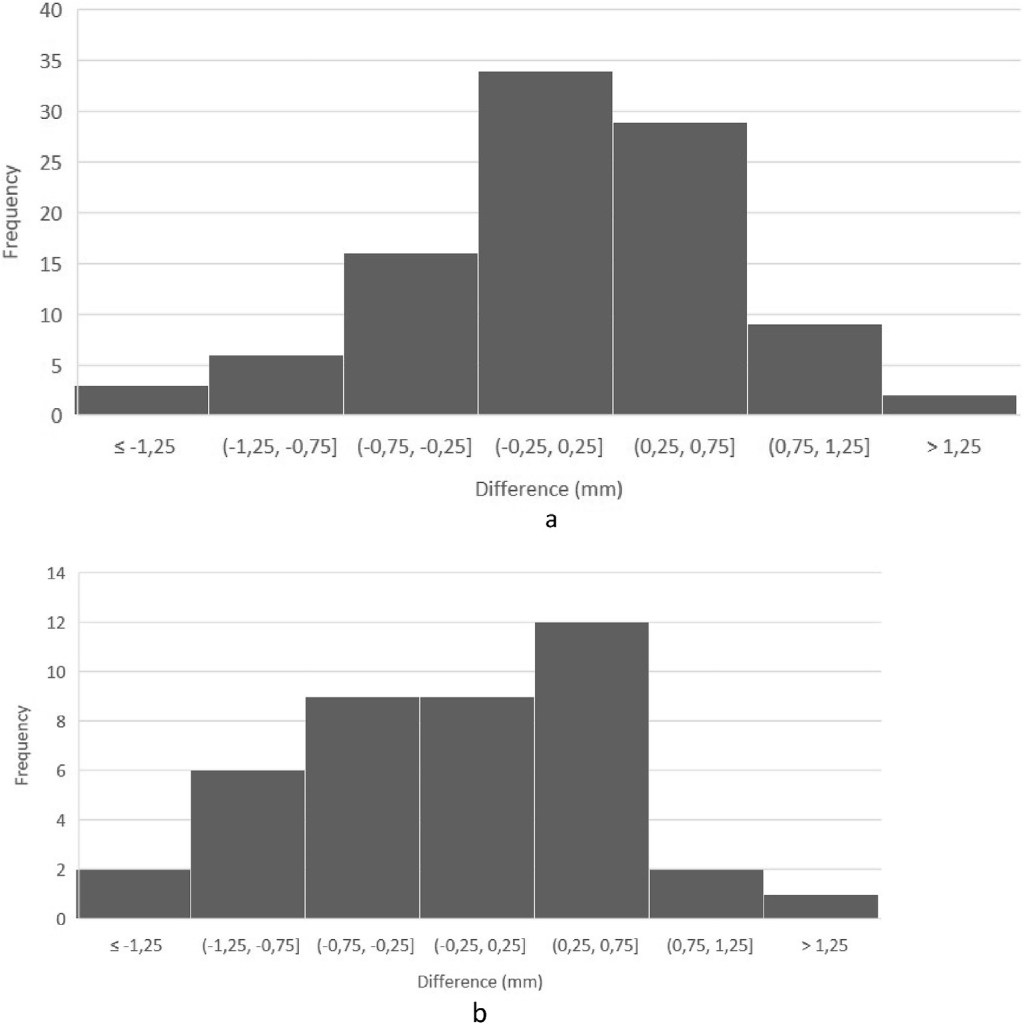
between the software and manual reporting on the perforators included in the flap.
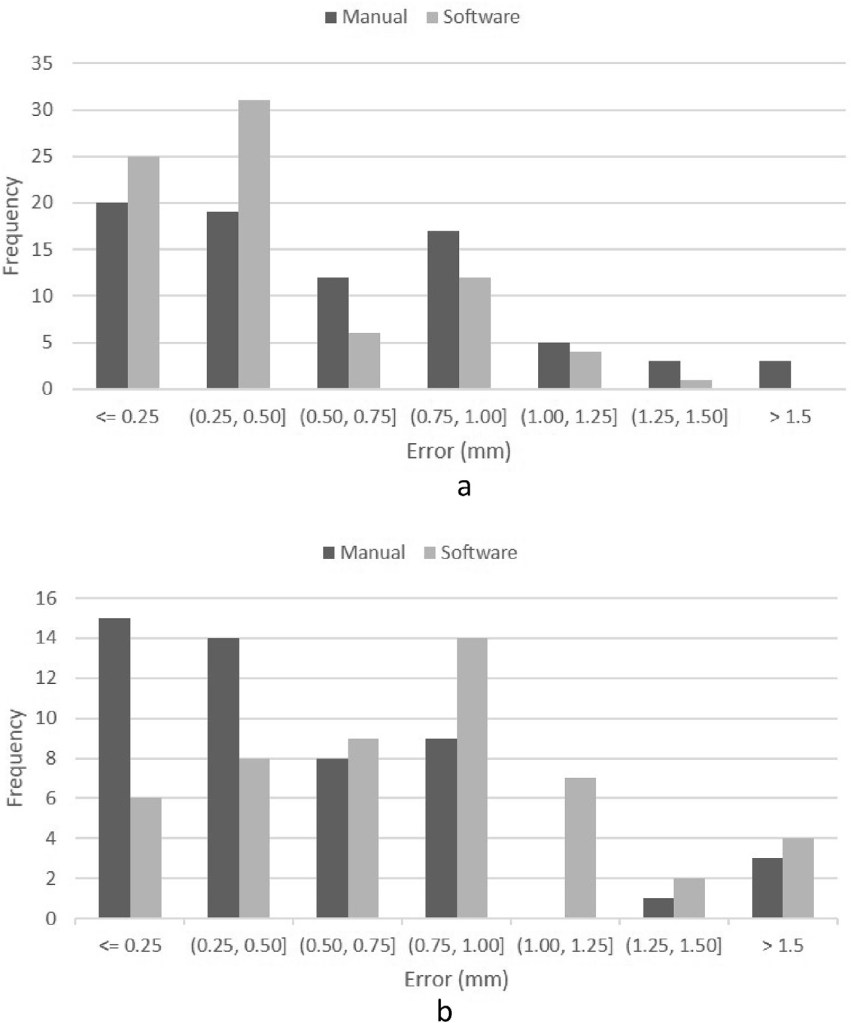
less or equal to 1,5 mm.
A statistically significant reduction of the median absolute error of estimated calibres was obtained by using the software, regarding perforators having calibres larger than 1.5 mm, P ≈ 2e-3, with a medium effect size, r = 0.26. However, using the software led to a statistically significant increase of the median absolute error when estimating the calibre of perforators having a calibre less or equal to 1,5 mm, P ≈ 6e-4. The effect size was medium, r = 0.34. Fig. 6 a and b shows the distribution of the absolute errors of both methods when comparing with the intraoperative measures of, respectively, perforators having calibre larger than 1.5 mm, and the ones having calibre less or equal to 1.5 mm.
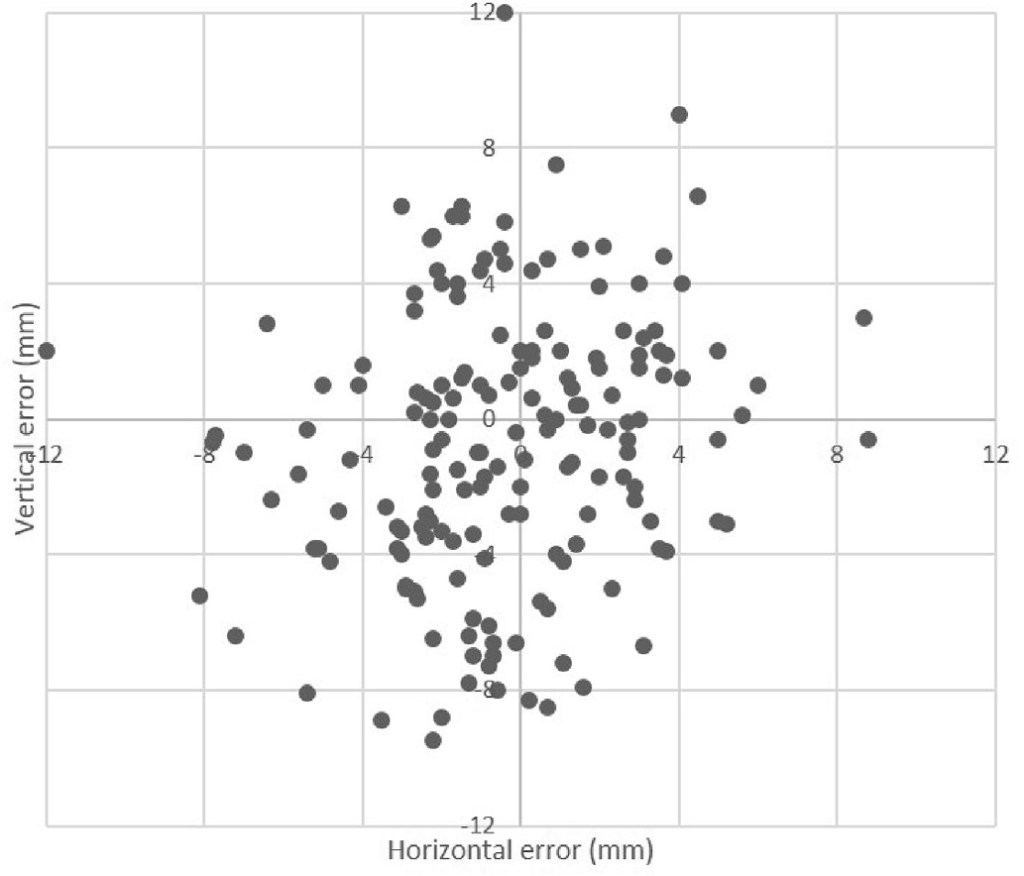
Fig. 7. Localization estimated by the software in comparison with the manual report.
Regarding the software estimates of where the perforators leave the fascia, horizontal error was not statistically significant, P ≈ 0.09 (170º of freedom), and the effect size was small, r = 0.13. However, vertical error was statistically significant, P ≈ 0.02 (170º of freedom), but the effect size was also small, r = 0.18. Fig. 7 shows a representation of the software localization estimates according to the manual reference. Regarding error in height, the average absolute error was 3.2 ± 2.4 mm, whereas the horizontal average absolute error was 2.5 ± 2.0 mm.
Considering the time taken to analyse the perforators of these 40 patients (2e3 h in the manual analysis and 30 min when using the software), approximately 80 h were saved using the semiautomatic methodology.
4. DISCUSSION
Although conservative treatment for breast cancer due to early detection and better neo-adjuvant treatments has raised impressively [10], mastectomy still has a role when the former is not possible and, in the case of high-risk patients, due to, most frequently, germline mutations in breast cancer predisposition genes. In these cases, immediate or delayed reconstruction should always be proposed, and autologous techniques using free tissue transfer are commonly chosen options.
Although microsurgery has already 50 years of existence, the last years have shown a wider range of surgical indications and a reduction of complications due to not only wider experience, but also to technological advances.
One of the most important advances is the possibility of using imaging techniques to pre-operatively identify the perforators that will be more suitable for a successful reconstruction. The most frequently performed imaging techniques are the Angio CT and the Angio MRI, allowing the creation of a map that will guide the surgeon in the process of identifying the best vessel(s) for the microsurgical reconstruction. Although the use of imaging techniques helps to reduce the duration of surgery [3], research on the use of these methods is scarce and several factors (type of machine, experience of the imaging team) can influence the final report.
The results allow to perceive some merits but also limitations of the semi-automatic approach [9] when compared with the manual analysis. It was more adequate at measuring the calibre of larger perforators (≥ 1.5 mm) but also shown a tendency to overestimate the calibre of the smallest ones. Regarding the localization of the perforators, even though there was a small error regarding vertical position (2e3 mm), it was not relevant in clinical practice, since perforators are approached carefully during dissection in order to prevent damage.
The majority (88%) of the perforators included in the flaps were co-identified by both methods, showing that the results of the semi-automatic methodology can be an option for this particular scenario. Despite having very challenging signal-to-noise ratio, the deep inferior epigastric vessel tree has a relatively simple network such that centreline-based approaches are the framework of choice amongst traditional algorithms for blood vessel segmentation. Additionally, such methodology also facilitates the local characterization of blood vessels, which is essential in this scenario. Although the proposed pipeline is not completely automatic, the effort required by the user is significantly reduced. Moreover, these algorithms make it easier for the user to interact with them in case errors are spotted. As they output a sequence of vessel centreline points, it is easy to manually manipulate the output to correct eventual mistakes.
Machine-learning based solutions, very commonly used nowadays in many applications, were not found particularly useful here yet, since example segmentations from experts are not available and obtaining a reasonable data set would be very time consuming. Even though there are some annotations specifying voxels belonging to vessels, the amount is not enough for a successful use of machine learning techniques. However, in the future, as new data keeps being accumulated and analysed with the semiautomatic approach [9], the outputs may start being used to learn models in a weakly supervised fashion. Thus, it is expected that such branch of machine-learning may be helpful to propose completely automatic algorithms in the future, possibly reducing the time of pre-operative analysis to a few seconds.
Results obtained with the Angio CT are expected to be mirrored in Angio MRI, as images obtained with the latter also show vessels with contrast, and usually with better detail. Since many units use Angio MRI due to less radiation exposure, it will make sense to also apply this algorithm to this setting in the future.
These results are promising in the sense that the semiautomatic methodology [9] was capable of delineating and characterizing the perforators with an adequate performance, reducing significantly the operator-dependent analysis (at least 2 h per patient) removing many of the time-consuming steps.
5. CONCLUSIONS
Imaging techniques are progressively associated with artificial intelligence approaches to try to improve detection accuracy, reproducibility and to decrease the time-consuming task of human input.
In the field of microsurgery, the use of imaging techniques to detect perforator vessels is routine, but all the process of annotation is, until now, manual and dependent on the imaging team.
In this preliminary work, we show that a semi-automatic algorithm reaches promising performance, with the advantage of significantly reducing the time consumed in the task. The difference came essentially from the almost instantaneous identification of the perforators, after receiving little input from the user.
The future will pass through enlarging the amount of data by including more patients, testing the algorithm with different imaging techniques (e.g. Angio MRI), and improving the process into a completely automatic tool that would ultimately be available in all imaging machines performing this kind of exam.
REFERENCES
[1] Allen RJ, Treece P. Deep inferior epigastric perforator flap for breast reconstruction. Ann Plast Surg 1994;32(1):32e8.
[2] Seidenstuecker K, van Waes C, Munder BI, Claes KE, Witzel C, Roche N, et al. DIEAP flap for safe definitive autologous breast reconstruction. Breast 2016;26:59e66.
[3] Wade RG, Watford J, Wormald JCR, Bramhall RJ, Figus A. Perforator mapping reduces the operative time of DIEP flap breast reconstruction: a systematic review and meta-analysis of preoperative ultrasound, computed tomography and magnetic resonance angiography. J Plast Reconstr Aesthet Surg 2018;71(4):468e77.
[4] Klasson S, Svensson H, Malm K, Wasselius J, Velander P. Preoperative CT angiography versus Doppler ultrasound mapping of abdominal perforator in DIEP breast reconstructions: a randomized prospective study. J Plast Reconstr Aesthet Surg 2015;68(6):782e6.
[5] Gomez-Cia T, Gacto-Sanchez P, Sicilia D, Suarez C, Acha B, Serrano C, et al. The virtual reality tool VirSSPA in planning DIEP microsurgical breast reconstruction. Int J Comput Assist Radiol Surg 2009;4(4):375e82.
[6] Lange CJ, Thimmappa ND, Boddu SR, Dutruel SP, Pei M, Farooq Z, et al. Automating perforator flap MRA and CTA reporting. J Digit Imaging 2017;30(3):350e7.
[7] Asl ME, Koohbanani NA, Frangi AF, Gooya A. Tracking and diameter estimation of retinal vessels using Gaussian process and Radon transform. J Med Imaging 2017;4(3):034006.
[8] Chae MP, Ganhewa D, Hunter-Smith DJ, Rozen WM. Direct augmented reality computed tomographic angiography technique (ARC): an innovation in preoperative imaging. Eur J Surg 2018;41(4):415e20.
[9] Araujo RJ, Garrido V, Baracas CA, Vasconcelos MA, Mavioso C, Anacleto JC, et al. Computer aided detection of deep inferior epigastric perforators in computed tomography angiography scans. Comput Med Imag Graph 2019;77: 101648.
[10] Gentilini OD, Cardoso MJ, Poortmans P. Less is more. Breast conservation might be even better than mastectomy in early breast cancer patients. Breast 2017;35:32e3.
